Understanding the Situation: Biopasteur is a biotechnology venture founded by three friends, Arnold Hand, Jeff Thompson and Amy Waitz in 2002. Biopasteur uses bio-technology to develop new cheaper, patient-friendly drugs to cure diseases. Biopasteur became profitable in 2006 majorly because of a pioneer blood pressure drug (LOBLOPRIN). BioPasteur plans to run till 2019 after which it is projected to cease operations.
Profit hit the $50m mark in 2011 despite a slow start and was expected to grow at a rate of 10% every year until 2019. DIASTOP was developed Between 2006 and 2011, a new drug to cure type 1 and type 2 diabetes and expected to be more profitable than LOBLOPRIN when introduced. DIASTOP was approved by the FDA despite 45 of 1000 human test subjects developing side effects and 3 of the subjects hospitalized.
Identifying and defining the problem: BioPasteur is having troubles with deciding on whether to release DIASTOP immediately or hold it back for further R&D. Jeff believes DIASTOP has potential for long-term negative effects on the users and advocate for more investments on R&D to possibly improve the product despite the likely loss of investment on costs already sunk into DIASTOP. However, the other two are of the opinion that DIASTOP’s tests results are satisfactory and should be released immediately.
Objective: The objective points towards making the best decisions concerning the release of DIASTOP without it harming patients whilst maintaining the company’s reputation.
Alternative solutions;
- Take DAISTOP into the market now.
- To not Launch DIASTOP.
- Invest in improving DIASTOP while delaying the release.
Criteria
Company reputation (recall threat),
Profitability,
Costs (R&D and Production),
Market expectations.
Timing of drug release.
Evaluation of alternatives
Company Reputation(Recall threat): Launching DIASTOP would be a positive on the company’s reputation. However, the impact of a failed launch of the new product would not only deter the market from DIASTOP but could also significantly impact on the sales of Lobloprin, the company’s pioneer product. Choosing to develop the new product further would ensure that the company’s image and reputation is maintained, and the success of its existing product not hampered. The release of the drug may have negative impacts on the reputation of the owners, their future plans and the company at large if there is a recall.
Profitability: The decision to release DIASTOP now would result in immediate revenue generation and profits for the firm which is estimated at about $70million yearly. If the product is delayed the earliest profits estimated at the same $70miilion would be made four years later and there is a 50% probability that the product may be released later than four years and may not yield any returns. Furthermore, a recall threat exists with the potential of damaging the profit performance of the already existing drug.
Cost: Deciding to release the product immediately would mean BioPasteur incurs no further costs asides the $15million expenses made for production. Should the company choose to defer the release of the product however BioPasteur would be committing to incur an additional $12million annually in research and development costs which have a probability of not being recouped.
Market expectations: In choosing to release the DIASTOP in its current state, BioPasteur would be meeting its market’s expectation in terms of the built-up anticipation which could yield positive results for the company.
Timing of drug release: Launching DIASTOP would be a positive for the company as they would harness the full benefits of the first mover advantage.
Estimated BioPasteur Profits projections for 4 scenarios (2012 – 2019)
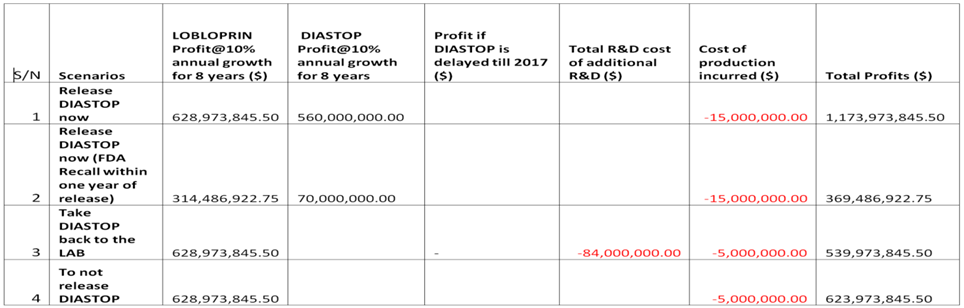
- Assumptions:
- No event of FDA recalls (all 8 years) if DIASTOP is launched immediately.
- FDA recall within one year of release/LOBLOPRIN’s profit is affected by 50%.
- 3/4. Production contract be revoked from GFT.
- * 2011 is far spent.
Decision
Considering profitability alone, the decision would be skewed more towards immediate release. However, BioPasteur, Being a more socially and ethically conscious organization, emphasis would be on reputation and regulatory concerns over profitability, costs and time. As such my recommendation would be for BioPasteur to stop any further developments on DIASTOP and terminate any launch into the market.
Action Plan. Immediately terminate any further developments or Launch on DIASTOP.
Evaluation of Action plan: BioPasteur can consider selling the patent to another competent pharmaceutical company for an outright price (Minimum valuation – $ 550,000,000.00).




 The Emergence of Artificial Intelligence: Shaping the Contemporary Landscape
The Emergence of Artificial Intelligence: Shaping the Contemporary Landscape